Intro
Effortlessly convert 1 atomic mass unit (amu) to kilograms (kg) with our simplified guide. Learn the exact conversion factor and understand the relationship between amu, unified atomic mass units (u), and kilogram-force (kgf). Master the 1 amu to kg conversion with ease and accuracy, perfect for students and professionals in chemistry and physics.
The world of physics and chemistry can be a complex and daunting place, especially when it comes to units of measurement. One such unit that is often used in atomic physics is the atomic mass unit (amu). However, when working with larger quantities, it's often necessary to convert amu to kilograms. In this article, we'll explore the concept of amu and kg, and provide a step-by-step guide on how to convert between the two.
The atomic mass unit (amu) is a unit of mass that is used to express the mass of atoms and molecules. It is defined as one-twelfth the mass of a carbon-12 atom, which is approximately equal to 1.66053904 × 10^-27 kilograms. The amu is commonly used in chemistry and physics to express the mass of small particles, such as atoms and molecules.
On the other hand, the kilogram (kg) is a unit of mass that is used to express larger quantities, such as the mass of everyday objects. It is defined as the mass of the International Prototype of the Kilogram (IPK), which is a platinum-iridium alloy cylinder that is stored at the International Bureau of Weights and Measures in France.
Converting between amu and kg can be a bit tricky, but don't worry, we've got you covered. In the next section, we'll provide a step-by-step guide on how to convert amu to kg.
Converting Amu to Kg: A Step-by-Step Guide

Converting amu to kg is a simple process that involves multiplying the amu value by a conversion factor. Here's a step-by-step guide:
- Write down the amu value that you want to convert.
- Multiply the amu value by the conversion factor: 1 amu = 1.66053904 × 10^-27 kg.
- The result is the kg value.
For example, let's say we want to convert 10 amu to kg.
10 amu × (1.66053904 × 10^-27 kg/amu) = 1.66053904 × 10^-26 kg
Therefore, 10 amu is equal to 1.66053904 × 10^-26 kg.
Practical Applications of Amu to Kg Conversion
Converting amu to kg has several practical applications in physics and chemistry. Here are a few examples:
- Calculating the mass of a molecule: When working with molecules, it's often necessary to calculate their mass in kg. By converting the amu value of the molecule to kg, we can determine its mass in a more familiar unit.
- Calculating the energy of a reaction: In chemistry, reactions involve the conversion of one substance to another. By converting the amu value of the reactants and products to kg, we can calculate the energy of the reaction.
- Calculating the density of a substance: Density is defined as mass per unit volume. By converting the amu value of a substance to kg, we can calculate its density in kg/m³.
Benefits of Converting Amu to Kg

Converting amu to kg has several benefits, including:
- Simplifying calculations: By converting amu to kg, we can simplify calculations and avoid working with very small or very large numbers.
- Improving accuracy: Converting amu to kg can improve the accuracy of calculations, especially when working with very small or very large quantities.
- Enhancing understanding: By converting amu to kg, we can gain a better understanding of the mass of atoms and molecules, and how they relate to everyday objects.
Common Mistakes to Avoid When Converting Amu to Kg
When converting amu to kg, there are several common mistakes to avoid, including:
- Forgetting to multiply by the conversion factor: Make sure to multiply the amu value by the conversion factor: 1 amu = 1.66053904 × 10^-27 kg.
- Rounding errors: Be careful when rounding numbers, as this can lead to errors in the calculation.
- Units confusion: Make sure to use the correct units, kg, and not other units such as g or mg.
Conclusion
In conclusion, converting amu to kg is a simple process that involves multiplying the amu value by a conversion factor. By following the steps outlined in this article, you can easily convert amu to kg and simplify your calculations. Remember to avoid common mistakes, such as forgetting to multiply by the conversion factor, rounding errors, and units confusion. With practice, you'll become proficient in converting amu to kg and be able to tackle complex calculations with ease.
Amu to Kg Conversion Image Gallery





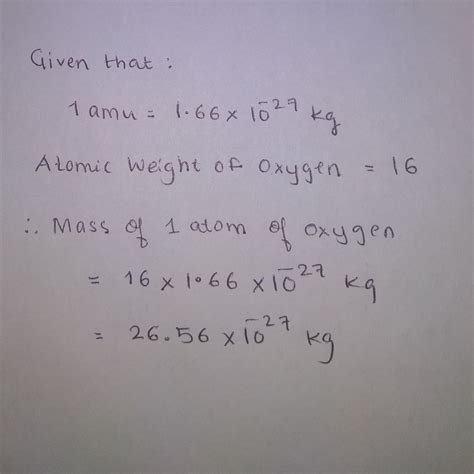


What is the conversion factor for amu to kg?
+The conversion factor for amu to kg is 1 amu = 1.66053904 × 10^-27 kg.
Why is it important to convert amu to kg?
+Converting amu to kg is important because it allows us to express the mass of atoms and molecules in a more familiar unit, and it simplifies calculations.
What are some common mistakes to avoid when converting amu to kg?
+Common mistakes to avoid when converting amu to kg include forgetting to multiply by the conversion factor, rounding errors, and units confusion.
